

The biphasic rTMS stimulator is designed for clinical and research applications, offering up to 100 pps, cooled coils, MEP integration, and a counter-balanced coil holder. The XTR supports TBS protocols at 100% stimulator intensity


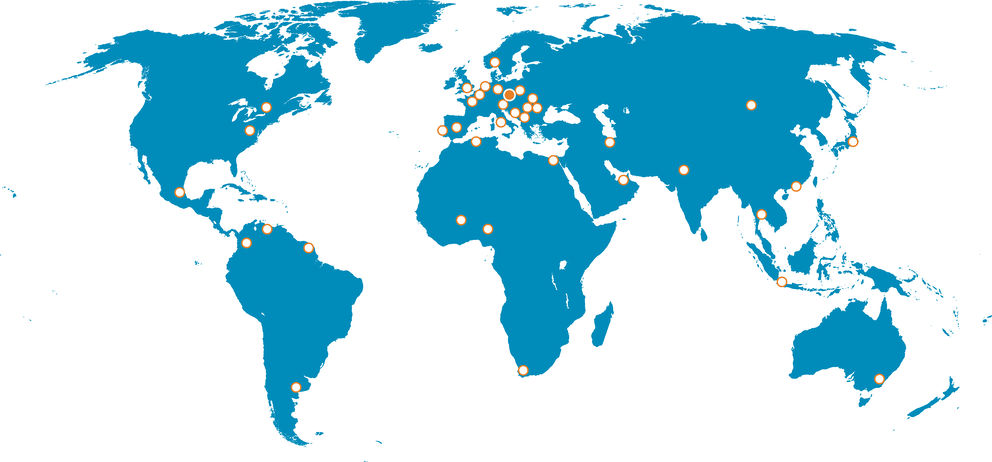
Where you can find our products
Years on the market
29Countries with DEYMED products
60+Products in the world
12000+DEYMED Diagnostic is a leading Czech company with an established global existence, specialising in advanced medical diagnostic and treatment technologies for neurophysiology and psychiatry. Founded in 1997, DEYMED has expanded its product range over the years to include EEG, EMG, TMS (Transcranial Magnetic Stimulation), PSG (Polysomnography), and BFB (Biofeedback) systems, trusted by healthcare providers worldwide for their precision, reliability, and ease of use.
Focusing on innovation and quality, DEYMED has become one of the leaders in both clinical and research applications. Its solutions are engineered for signal quality, multifunctionality and reliability with in-house development ensuring that all products meet healthcare standards. Each manufactured device undergoes strict testing to ensure safety and performance.
Through ongoing investment in R&D, DEYMED is pushing the boundaries of innovation in neurophysiology and psychiatry, improving patient outcomes and making progress in the field.
Watch our video and learn more about the company.
During the development process we closely cooperate with leading healthcare professionals for insights and feedback into the features and design of our products. This helps us be sure that we are always meeting the most demanding requirements of our customers, as well as helping us keep an eye out for opportunities to innovate by using the latest in technology to even exceed the expectations of our users.
Our goal is to develop products that meet and exceed the highest standards in neurology and psychiatry. To help meet this standard, all our products are designed and developed according to ISO 13485 and FDA 21 CFR part 820 requirements of quality systems.
Our devices meet strict safety requirements for medical devices according to European standards EN 60601. Compliance with safety and regulatory requirements for the marketing of our products is demonstrated by their CE marking.
We offer our customers above-standard care in technical and information support. Our professional team provides fast and efficient customer service as well as remote support using state-of-the-art communication tools.
Download Quick Remote Support App
Quick Support is better for single support events when you will be present when the tech connects.
Download Unattended Remote Support App
Unattended Support is better for support events when you will not be present and/or the tech might need to connect more than one time.
After launching Quick Support, please contact us at support@deymed.com or by phone. Our technician will provide you with a one-time connection code. If you download Unattended Support, an e-mail containing the completed information will be automatically sent to our support team, who will then contact you.
Other helpful links:
» Minimum System Requirements for DEYMED systems
DEYMED Systems are designed to run on any standard Windows Desktop or Laptop that meet the following minimum requirements:
TruScan, SomniPro:
TruTrace EMG:
Neurofeedback:
Troubleshooting Resolution Issues:
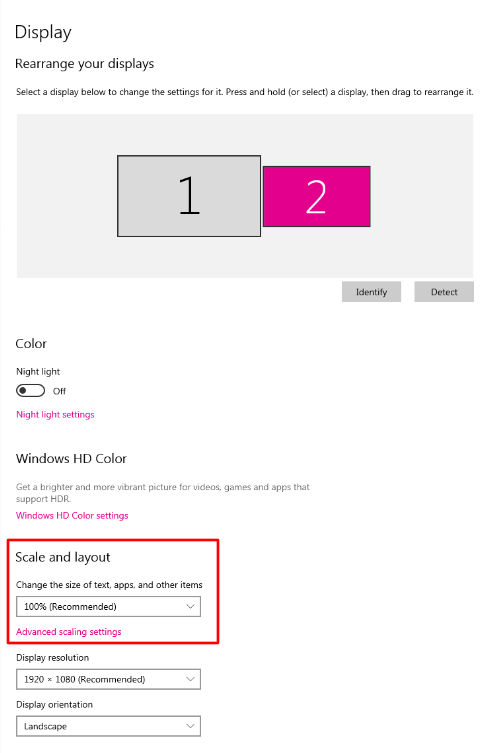
Some recent versions of Windows have a setting that increases the size of screen elements. This setting can cause problems in a Dashboard style interface like DEYMED’s by making items larger than they were designed. This can cause some elements to be pushed off the screen. To correct this, be sure there is no DPI setting above 100% or other scalling setting over 100% in the display settings. This setting is typically found in the Advanced Display Settings. In Windows 10, the setting looks like this:
For support, please call: +420 491 481 038
To log a support case with us, please login to our customer ePortal at the top of this page.
If you do not have an account, please register.
Billing address:
Kudrnáčova 533
549 31 Hronov
REG. ID: 25284584
VAT ID: CZ25284584
Shipping address:
Kudrnáčova 533
549 31 Hronov
Registered at the Regional Court in Hradec Kralove, Section C, File no. 12882